CAS No.: 941678-49-5
芦可替尼 Catalog No. CSN12394
Synonyms: INCB18424;INCB 018424;INC-424
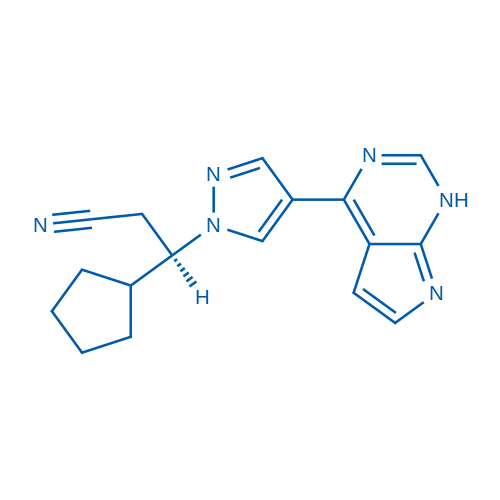
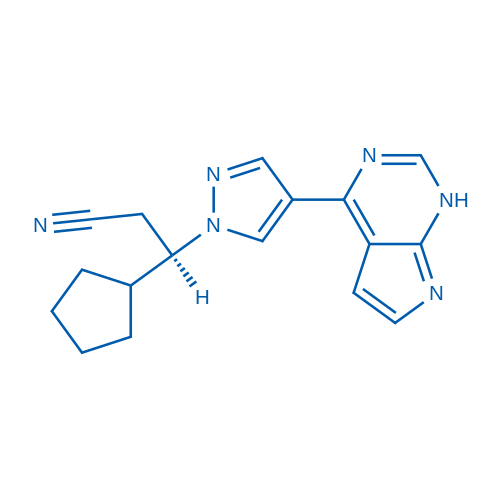
Ruxolitinib is the first potent, selective, JAK1/2 inhibitor to enter the clinic with IC50 of 3.3 nM/2.8 nM, > 130-fold selectivity for JAK1/2 versus JAK3.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 JAK1
IC50:3.3nMJAK2
IC50:2.8nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT03274778 Prostate Cancer Not Applicable Recruiting May 1, 2019 Switzerland ... more >> Istituto Oncologico della Svizzera Italiana (IOSI) Recruiting Bellinzona, Switzerland, 6500 Contact: Ricardo Pereira Mestre, Dr. med. +41 (0)91 811 84 46 Ricardo.PereiraMestre@eoc.ch Contact: Barbara Marongiu +41 (0)91 811 91 20 barbara.marongiu@eoc.ch Collapse << NCT02120417 - - Terminated(The study was termi... more >>nated as other related studies of ruxolitinib did not provide sufficient efficacy to warrant continuation.) Collapse << - - NCT01914484 Chronic Phase Chronic Myeloid ... more >>Leukemia Accelerated Phase Chronic Myeloid Leukemia Blastic Phase Chronic Myeloid Leukemia Philadelphia Positive Acute Lymphoblastic Leukemia Resistant to Tyrosine Kinase Inhibitor Therapy Collapse << Phase 1 Phase 2 Unknown July 2016 Canada, Ontario ... more >> Princess Margaret Hospital / University Health Network Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Sima Bogomilsky, RN BScN CON(C) 416-946-4646 sima.bogomilsky@uhn.on.ca Contact: Sonal Malhotra, M.Sc., Ph.D,CCRP 416-946-4501 ext 3449 sonal.malhotra@uhn.ca Principal Investigator: Dennis Kim, MD/PhD Sub-Investigator: Jeffrey H Lipton, MD/PhD Collapse << - 更多
- 参考文献
- [1] Quint¨¢s-Cardama A, Vaddi K, et al. Preclinical characterization of the selective JAK1/2 inhibitor INCB018424: therapeutic implications for the treatment of myeloproliferative neoplasms. Blood. 2010 Apr 15;115(15):3109-17.
- [2] Hobbs GS, Rozelle S, et al. The Development and Use of Janus Kinase 2 Inhibitors for the Treatment of Myeloproliferative Neoplasms. Hematol Oncol Clin North Am. 2017 Aug;31(4):613-626.
- [3] Heine A, Held SA, et al. The JAK-inhibitor ruxolitinib impairs dendritic cell function in vitro and in vivo. Blood. 2013 Aug 15;122(7):1192-202.
- [4] Silvennoinen O, Hubbard SR, et al. Targeting the Inactive Conformation of JAK2 in Hematological Malignancies. Cancer Cell. 2015 Jul 13;28(1):1-2
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 941678-49-5 | 储存条件 |
|
|||||||
| 分子式 | C17H18N6 | 运输 | 蓝冰 | |||||||
| 分子量 | 306.37 | 别名 | INCB18424;INCB 018424;INC-424;trade name:J akafi and Jakavi;芦可替尼;鲁索替尼 | |||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| A549 | - | Growth Inhibition Assay | - | IC50=0.04 μM | 25213670 |
| A549/DDP | - | Growth Inhibition Assay | - | IC50=0.22 μM | 25213670 |
| A549/DDP | 30 nM | Function Assay | 48 h | Down-regulation of STAT3 phosphorylation | 25213670 |
| A549/DDP | 30 nM | Apoptosis Assay | 48 h | Induction of apoptosis | 25213670 |
| BaF3 | 80 nM | Kinase Assay | 6 h | Reduces the phosphorylation of STAT5 in JAK2V617F-mutated BAF3-EPOR cell | 24237791 |
| CMK | - | Growth Inhibition Assay | - | Inhibition of CMK carrying the JAK3A572V mutation cell proliferation | 25352124 |
| CMK | - | Growth Inhibition Assay | - | Inhibition of CMK carrying the JAK3A63D mutation cell proliferation with IC50 of 0.163 μM | 25352124 |
| CMK | - | Growth Inhibition Assay | - | Inhibition of CMK carrying the WT JAK cell proliferation with IC50 of 0.075 μM | 25352124 |
| DLD-1 | 25 μM | Kinase Assay | 48 h | Inhibition of JAK1 phosphorylation | 24050550 |
| DLD-1 | 25 μM | Kinase Assay | 48 h | Inhibition of JAK2 phosphorylation | 24050550 |
| DLD-1 | 50 μM | Growth Inhibition Assay | 48 h | IC50=15.51 μM | 24050550 |
| DLD-1 | 25 μM | Apoptosis Assay | 48 h | Induces apoptosis by activating caspase 3 | 24050550 |
| HEL | 5 μM | Cytotoxic Assay | 48 h | Cytotoxic index=12.2% | 25931349 |
| Hep3B | 1 μM | Function Assay | 16 h | Impaires the capacity of IHCA-associated gp130 mutants to active STAT3 with IC50 of ~50 μM | 24501689 |
| HepG2 | 1 μM | Function Assay | 16 h | Impaires the capacity of IHCA-associated gp130 mutants to signal to STAT3 | 24501689 |
| HT93A | 320 nM | Growth Inhibition Assay | 5 d | Inhibition of GCS-F induced granulocytic differentiation | 25805962 |
| Huh7 | 1 μM | Function Assay | 16 h | Impaires the capacity of IHCA-associated gp130 mutants to signal to STAT3 | 24501689 |
| HuH7 | 50 μM | Growth Inhibition Assay | 48 h | >82% reduction | 23941832 |
| HuH7 | 50 μM | Function Assay | 24 h | Inhibition of STAT1 and STAT3 phosphorylation significantly | 23941832 |
| Human monocyte | - | Kinase Assay | - | Inhibition of JAK2 in human monocytes expressing CD14 assessed as inhibition of GM-CSF-stimulated STAT5a phosphorylation with IC50 of 0.026μM | 23540648 |
| Human monocyte | - | Kinase Assay | - | Inhibition of JAK2/1 in human monocytes expressing CD14 assessed as inhibition of IFNgamma-stimulated STAT1 phosphorylation with IC50 of 0.031μM | 23540648 |
| Human T cell | - | Kinase Assay | - | Inhibition of JAK3/1 in human T cells expressing CD3 assessed as inhibition of IL2-stimulated STAT5a phosphorylation with IC50 of 0.023μM | 23540648 |
| NCI-H1299 | - | Growth Inhibition Assay | - | IC50=0.28 μM | 25213670 |
| NCI-H1299 | 30 nM | Function Assay | 48 h | Down-regulation of STAT3 phosphorylation | 25213670 |
| NCI-H1299 | 30 nM | Apoptosis Assay | 48 h | Induction of apoptosis | 25213670 |
| NCI-H2347 | - | Growth Inhibition Assay | - | IC50=0.17 μM | 25213670 |
| NCI-H2347 | 30 nM | Function Assay | 48 h | Decrease in Bcl2 expression | 25213670 |
| NCI-H2347 | 30 nM | Apoptosis Assay | 48 h | Induction of apoptosis | 25213670 |
| NCI-H358 | - | Growth Inhibition Assay | - | IC50=0.1 μM | 25213670 |
| NCI-H460 | - | Growth Inhibition Assay | - | IC50=0.13 μM | 25213670 |
| RKO | 25 μM | Kinase Assay | 48 h | Inhibition of JAK1 phosphorylation | 24050550 |
| RKO | 25 μM | Kinase Assay | 48 h | does not inhibit JAK1 phosphorylation | 24050550 |
| RKO | 50 μM | Growth Inhibition Assay | 48 h | IC50=14.76 μM | 24050550 |
| RKO | 25 μM | Apoptosis Assay | 48 h | Induces apoptosis by activating caspase 3 | 24050550 |
| SET-2 | 5 μM | Cytotoxic Assay | 48 h | Cytotoxic index=18.7% | 25931349 |
| SNU182 | 50 μM | Growth Inhibition Assay | 48 h | >64% reduction | 23941832 |
| SNU182 | 50 μM | Function Assay | 24 h | Inhibition of STAT1 and STAT3 phosphorylation significantly | 23941832 |
| SNU423 | 50 μM | Growth Inhibition Assay | 48 h | >81% reduction | 23941832 |
| SNU423 | 50 μM | Function Assay | 24 h | Inhibition of STAT1 and STAT3 phosphorylation significantly | 23941832 |
| TF1 | - | Kinase Assay | 20 min | Inhibition of JAK2 in human TF1 cells assessed as inhibition of EPO-induced STAT5 phosphorylation with IC50 of 0.012μM | 22698084 |
| TF1 | - | Kinase Assay | 20 min | Inhibition of JAK1 in human TF1 cells assessed as inhibition of IL6-induced STAT3 phosphorylation with IC50 of 0.024μM | 22698084 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT03274778 | Prostate Cancer | Not Applicable | Recruiting | May 1, 2019 | Switzerland ... more >> Istituto Oncologico della Svizzera Italiana (IOSI) Recruiting Bellinzona, Switzerland, 6500 Contact: Ricardo Pereira Mestre, Dr. med. +41 (0)91 811 84 46 Ricardo.PereiraMestre@eoc.ch Contact: Barbara Marongiu +41 (0)91 811 91 20 barbara.marongiu@eoc.ch Collapse << |
| NCT02120417 | - | - | Terminated(The study was termi... more >>nated as other related studies of ruxolitinib did not provide sufficient efficacy to warrant continuation.) Collapse << | - | - |
| NCT01914484 | Chronic Phase Chronic Myeloid ... more >>Leukemia Accelerated Phase Chronic Myeloid Leukemia Blastic Phase Chronic Myeloid Leukemia Philadelphia Positive Acute Lymphoblastic Leukemia Resistant to Tyrosine Kinase Inhibitor Therapy Collapse << | Phase 1 Phase 2 | Unknown | July 2016 | Canada, Ontario ... more >> Princess Margaret Hospital / University Health Network Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Sima Bogomilsky, RN BScN CON(C) 416-946-4646 sima.bogomilsky@uhn.on.ca Contact: Sonal Malhotra, M.Sc., Ph.D,CCRP 416-946-4501 ext 3449 sonal.malhotra@uhn.ca Principal Investigator: Dennis Kim, MD/PhD Sub-Investigator: Jeffrey H Lipton, MD/PhD Collapse << |
| NCT02119650 | - | - | Terminated(The study was termi... more >>nated as other related studies of ruxolitinib did not provide sufficient efficacy to warrant continuation.) Collapse << | - | - |
| NCT03257644 | Atopic Dermatitis | Phase 1 | Suspended(Study is currently o... more >>n hold pending amendment update.) Collapse << | December 2019 | United States, California ... more >> Children's Hospital of Los Angeles Los Angeles, California, United States, 90027 Rady Children's Hospital - San Diego San Diego, California, United States, 92123 United States, Colorado National Jewish Health Denver, Colorado, United States, 80206 United States, Illinois Northwestern University Department of Dermatology Chicago, Illinois, United States, 60611 United States, Michigan David Fivenson, MD, Dermatology, PLC Ann Arbor, Michigan, United States, 48103 United States, North Carolina Wake Research Associates Raleigh, North Carolina, United States, 27612 United States, Pennsylvania Penn State Hershey Medical Center Hershey, Pennsylvania, United States, 17033 United States, Texas Texas Dermatology and Laser Specialist Clinical Research San Antonio, Texas, United States, 78218 Collapse << |
| NCT03654768 | Chronic Phase Chronic Myelogen... more >>ous Leukemia, BCR-ABL1 Positive Collapse << | Phase 2 | Recruiting | January 1, 2026 | United States, Oregon ... more >> SWOG Recruiting Portland, Oregon, United States, 97239 Contact: Kendra L. Sweet 813-745-6841 kendra.sweet@moffitt.org Principal Investigator: Kendra L. Sweet Collapse << |
| NCT02593760 | Myelofibrosis | Phase 1 | Completed | - | United States, Florida ... more >> Florida Cancer Specialists-Broadway, Fort Myers Fort Myers, Florida, United States, 33908 Florida Cancer Specialist, North Region Saint Petersburg, Florida, United States, 33705 Florida Cancer Specialists West Palm Beach, Florida, United States, 33401 United States, Ohio Oncology Hematology Care Inc Cincinnati, Ohio, United States, 45242 United States, Tennessee Sarah Cannon Research Institute Nashville, Tennessee, United States, 37203 United States, Texas Uni of Texas - Md Anderson Cancer Center; Dept of Leukemia Houston, Texas, United States, 77030 Canada, Alberta Tom Baker Cancer Centre-Calgary; Clinical Research Unit Calgary, Alberta, Canada, T2N 4N2 Canada, Nova Scotia Queen Elizabeth II Health Sciences Centre; Oncology Halifax, Nova Scotia, Canada, B3H 2Y9 Canada, Quebec Centre Hospitalier De L'Universite De Montreal, Hopital Notre-Dame Montreal, Quebec, Canada, H2L 4M1 Germany Uniklinik RWTH Aachen; Med. Klinik IV; Klinik für Hämatologie, Onkologie, Hämostaseologie und Stammz Aachen, Germany, 52074 Campus Virchow-Klinikum Charité Centrum 14; Medizinische Klinik m.S. Hämatologie u. Onkologie Berlin, Germany, 13353 Italy A.O.U. Citta' Della Salute E Della Scienza-P.O. Molinette;S.C. Ematologia Torino, Piemonte, Italy, 10126 Az. Osp. Di Careggi; Divisione Di Ematologia Firenze, Toscana, Italy, 50135 Collapse << |
| NCT01433445 | Idiopathic Myelofibrosis ... more >> Post Essential Thrombocythemia Myelofibrosis Post Polycythemia-Vera Myelofibrosis Collapse << | Phase 1 | Active, not recruiting | December 31, 2018 | France ... more >> Novartis Investigative Site Paris, France, 75010 Novartis Investigative Site Villejuif Cedex, France, 94805 Germany Novartis Investigative Site Magdeburg, Germany, 39120 Novartis Investigative Site Mainz, Germany, 55131 Ireland Novartis Investigative Site Dublin, Ireland, DUBLIN 8 Novartis Investigative Site Galway, Ireland Italy Novartis Investigative Site Firenze, FI, Italy, 50134 Novartis Investigative Site Reggio Calabria, RC, Italy, 89124 Novartis Investigative Site Varese, VA, Italy, 21100 United Kingdom Novartis Investigative Site London, United Kingdom, SE1 9RT Collapse << |
| NCT02494882 | Acute Lymphoblastic Leukemia | Phase 1 | Active, not recruiting | June 2019 | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01712659 | T Cell Leukemia, Adult ... more >> Leukemia, Adult T-Cell T Cell Leukemia, HTLV I Associated Collapse << | Phase 2 | Recruiting | November 1, 2021 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT03558607 | Secondary Acute Myelogenous Le... more >>ukemia Evolving From Myeloproliferative Disorder Collapse << | Phase 1 Phase 2 | Recruiting | December 31, 2021 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seongnam, Korea, Republic of, 13620 Contact: Ryul Kim, Dr +82 10 9412 6108 chrono0707@icloud.com Seoul National University Hospital Recruiting Seoul, Korea, Republic of, 03080 Contact: Ryul Kim, MD +82 10 9412 6108 chrono0707@icloud.com Principal Investigator: Youngil Koh, MD Sub-Investigator: Sung soo Yoon, MD, PhD Sub-Investigator: In ho Kim, MD Sub-Investigator: Ryul Kim, MD Collapse << |
| NCT00952289 | MPN (Myeloproliferative Neopla... more >>sms) Collapse << | Phase 3 | Completed | - | - |
| NCT02092324 | Atypical Chronic Myeloid Leuke... more >>mia, BCR-ABL1 Negative Chronic Neutrophilic Leukemia Collapse << | Phase 2 | Recruiting | - | United States, California ... more >> Stanford Cancer Institute Palo Alto Recruiting Palo Alto, California, United States, 94304 Contact: Jason Gotlib 650-736-1253 gotlib@stanford.org Principal Investigator: Jason Gotlib United States, Georgia Emory University Hospital/Winship Cancer Institute Recruiting Atlanta, Georgia, United States, 30322 Contact: Elliott F. Winton 404-778-4755 ewinton@emory.edu Principal Investigator: Elliott F. Winton United States, Missouri Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Stephen Oh 314-362-8846 stoh@dom.wustl.edu Principal Investigator: Stephen Oh United States, Oregon OHSU Knight Cancer Institute Recruiting Portland, Oregon, United States, 97239 Contact: Kim-Hien Dao 503-494-7894 daok@ohsu.edu Principal Investigator: Kim-Hien Dao United States, Texas UT Southwestern/Simmons Cancer Center-Dallas Recruiting Dallas, Texas, United States, 75390 Contact: Robert H. Collins 214-648-4155 robert.collins@utsouthwestern.edu Principal Investigator: Robert H. Collins M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Jorge E. Cortes 713-794-5783 jcortes@mdanderson.org Principal Investigator: Jorge E. Cortes United States, Utah Huntsman Cancer Institute/University of Utah Recruiting Salt Lake City, Utah, United States, 84112 Contact: Michael W. Deininger 801-213-5684 michael.deininger@hci.utah.edu Principal Investigator: Michael W. Deininger Collapse << |
| NCT00952289 | - | - | Completed | - | - |
| NCT01730755 | - | - | - | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT02420717 | Leukemia | Phase 2 | Recruiting | July 2023 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Collapse << |
| NCT02119663 | - | - | Terminated(The safety committe... more >>e found no safety issues but recommended halting the study based on a lack of efficacy in a similar trial. The sponsor terminated the trial.) Collapse << | - | - |
| NCT02119650 | NSCLC (Non-small Cell Lung Car... more >>cinoma) Collapse << | Phase 2 | Terminated(The study was termi... more >>nated as other related studies of ruxolitinib did not provide sufficient efficacy to warrant continuation.) Collapse << | - | - |
| NCT03774082 | Graft vs Host Disease | Phase 2 | Not yet recruiting | June 13, 2025 | - |
| NCT02117479 | Pancreatic Cancer | Phase 3 | Terminated(The study was termi... more >>nated early based on the results of the planned interim analysis.) Collapse << | - | - |
| NCT02974647 | Lymphoma | Phase 2 | Recruiting | November 2019 | United States, Florida ... more >> University of Miami Recruiting Miami, Florida, United States Contact: Jonathan Schatz, MD 305-689-5511 United States, Massachusetts Dana Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02115 Contact: Eric Jacobsen, MD 617-582-9086 United States, New Jersey Memorial Sloan Kettering Basking Ridge Recruiting Basking Ridge, New Jersey, United States, 07920 Contact: Alison Moskowitz, MD 212-639-4839 United States, New York Memorial Sloan Kettering Commack Recruiting Commack, New York, United States, 11725 Contact: Alison Moskowitz, MD 212-639-4839 Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Alison Moskowitz, MD 212-639-4839 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Alison Moskowitz, MD 212-639-4839 Contact: Steven Horwitz, MD 212-639-3045 Principal Investigator: Allison Moskowitz, MD Weill Cornell Medical Center Recruiting New York, New York, United States Contact: Jia Ruan, MD 646-962-2064 Principal Investigator: Jia Ruan, MD Collapse << |
| NCT02117479 | - | - | Terminated(The study was termi... more >>nated early based on the results of the planned interim analysis.) Collapse << | - | - |
| NCT03333187 | Bone Marrow Fibrosis | Phase 2 | Recruiting | March 20, 2023 | Germany ... more >> Universitätsklinkum Aachen Recruiting Aachen, Germany, 52074 Contact: Edgar Jost, PD Dr. ejost@ukaachen.de Principal Investigator: Edgar Jost, PD Dr. Sub-Investigator: Martina Crysandt, Dr. HELIOS Klinikum Berlin-Buch Recruiting Berlin, Germany, 13125 Contact: Herrad Baurmann, Dr. herrad.baurmann@helios-kliniken.de Principal Investigator: Herrad Baurmann, Dr. Sub-Investigator: Stephan Fuhrmann, Dr. Universitätsklinikum Bonn Recruiting Bonn, Germany, 53105 Contact: Dominik Wolf, Prof. Dr. dominik.wolf@ukb.uni-bonn.de Principal Investigator: Dominik Wolf, Prof. Dr. Sub-Investigator: Karin Mayer, Dr. Universitätsklinikum Düsseldorf Recruiting Düsseldorf, Germany, 40225 Contact: Guido Kobbe, Prof. Dr. Kobbe@med.uni-duesseldorf.de Principal Investigator: Guido Kobbe, Prof. Dr. Sub-Investigator: Thomas Schröder, PD Dr. Universitätsklinikum Essen Recruiting Essen, Germany, 45147 Contact: Joachim Göthert, Dr. Joachim.Goethert@uk-essen.de Principal Investigator: Joachim Göthert, Dr. Sub-Investigator: Jürgen Novotny, PD Dr. Klinikum Frankfurt (Oder) GmbH Not yet recruiting Frankfurt (Oder), Germany, 15236 Contact: Michael Kiehl, Prof. Dr. Contact: Olaf Hopfer, Dr. Principal Investigator: Olaf Hopfer, Dr. Sub-Investigator: Georgios Fellas Universitätsklinkum Halle Recruiting Halle (Saale), Germany, 06120 Contact: Haifa Kathrin Al-Ali, PD Dr. haifa.al-ali@uk-halle.de Principal Investigator: Haifa Kathrin Al-Ali, PD Dr. Sub-Investigator: Nadja Jäkel, Dr. University Medical Center Hamburg-Eppendorf Recruiting Hamburg, Germany, 20246 Contact: Nicolaus Kroeger, Prof. Dr. +49-40-7410-55864 n.kroeger@uke.de Contact: Marion Heinzelmann +49-40-7410-54188 mheinzel@uke.de Principal Investigator: Nicolaus Kroeger, Prof. Dr. Sub-Investigator: Christine Wolschke, Dr. Universitätsklinikum Jena Recruiting Jena, Germany, 07747 Contact: Inken Hilgendorf, PD Dr. Inken.Hilgendorf@med.uni-jena.de Principal Investigator: Inken Hilgendorf, PD Dr. Sub-Investigator: Nils Winkelmann, Dr. University Hospital Leipzig Recruiting Leipzig, Germany, 04103 Contact: Dietger Niederwieser, Prof. Dr. Dietger.Niederwieser@medizin.uni-leipzig.de Principal Investigator: Dietger Niederwieser, Prof. Dr. Sub-Investigator: Vladan Vucinic, Dr. Universitätsmedizin der Johannes Gutenberg-Universität Mainz Recruiting Mainz, Germany, 55131 Contact: Eva Wagner, Dr. eva.wagner@unimedizin-mainz.de Principal Investigator: Eva Wagner-Drouet, Dr. Sub-Investigator: Thomas Kindler, PD Dr. Universitätsmedizin Mannheim Recruiting Mannheim, Germany, 68167 Contact: Stefan Klein, PD Dr. Stefan.Klein@medma.uni-heidelberg.de Principal Investigator: Stefan Klein, PD Dr. Sub-Investigator: Andreas Reiter, Prof. Dr. Johannes Wesling Klinikum Minden Recruiting Minden, Germany, 32429 Contact: Martin Griesshammer, Prof. Dr. martin.griesshammer@muehlenkreiskliniken.de Principal Investigator: Martin Griesshammer, Prof. Dr. Sub-Investigator: Hans-Joachim Tischler, Dr. Universitätsklinikum Münster Recruiting Munster, Germany, 48149 Contact: Matthias Stelljes, Prof. Dr. Matthias.Stelljes@ukmuenster.de Principal Investigator: Eva Eßeling, Dr. Sub-Investigator: Matthias Stelljes, Prof. Dr. Klinikum Nürnberg Recruiting Nürnberg, Germany, 90419 Contact: Kerstin Schäfer-Eckart, Dr. Kerstin.Schaefer-Eckart@klinikum-nuernberg.de Principal Investigator: Kerstin Schäfer-Eckart, Dr. Sub-Investigator: Knut Wendelin, Dr, Robert-Bosch-Krankenhaus Stuttgart Recruiting Stuttgart, Germany, 70376 Contact: Martin Kaufmann, Dr. Martin.Kaufmann@rbk.de Principal Investigator: Martin Kaufmann, Dr. Sub-Investigator: Matthias Vöhringer, Dr. Universitätsmedizin Tübingen Recruiting Tübingen, Germany, 72076 Contact: Wolfgang Bethge, Prof. Dr. Wolfgang.Bethge@med.uni-tuebingen.de Principal Investigator: Wolfgang Bethge, Prof. Dr. Sub-Investigator: Christoph Faul, Dr. Universitätsklinkum Ulm Recruiting Ulm, Germany, 89081 Contact: Konstanze Döhner, Prof. Dr. Konstanze.Doehner@uniklinik-ulm.de Principal Investigator: Konstanze Döhner, Prof. Dr. Sub-Investigator: Frank Stegelmann, Dr. Collapse << |
| NCT02119663 | Pancreatic Cancer | Phase 3 | Terminated(The safety committe... more >>e found no safety issues but recommended halting the study based on a lack of efficacy in a similar trial. The sponsor terminated the trial.) Collapse << | - | - |
| NCT03386214 | Myelofibroses | Phase 1 | Recruiting | March 31, 2021 | United States, Missouri ... more >> Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Stephen Oh, M.D., Ph.D. 314-747-7960 stoh@wustl.edu Principal Investigator: Stephen Oh, M.D., Ph.D. Sub-Investigator: Amy Zhou, M.D. Collapse << |
| NCT01317875 | Myelofibrosis | Phase 1 | Active, not recruiting | September 2020 | United States, Florida ... more >> Winter Park, Florida, United States, 32789 United States, Maryland Baltimore, Maryland, United States, 21229 United States, Texas Houston, Texas, United States, 77030 Austria Vienna, Austria China, Jiangsu Nanjing, Jiangsu, China China, Sichuan Chengdu, Sichuan, China China, Zhejiang Hangzhou, Zhejiang, China China Beijing, China France Angers, France Paris, France Pierre-Benite, France Germany Leipzig, Germany Italy Firenze, Italy Milano, Italy Terni, Italy Netherlands Rotterdam, Netherlands United Kingdom Belfast, United Kingdom London, United Kingdom Collapse << |
| NCT02917096 | Primary Myelofibrosis ... more >> Secondary Myelofibrosis Collapse << | Not Applicable | Recruiting | October 2019 | United States, California ... more >> City of Hope Medical Center Recruiting Duarte, California, United States, 91010 Contact: Haris Ali 626-256-4673 Principal Investigator: Haris Ali Collapse << |
| NCT01375140 | Myeloproliferative Diseases | Phase 2 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02087059 | Primary Myelofibrosis (MF) | Phase 3 | Completed | - | Japan ... more >> Novartis Investigative Site Nagoya-city, Aichi, Japan, 467-8602 Novartis Investigative Site Matsuyama, Ehime, Japan, 790-8524 Novartis Investigative Site Toon-city, Ehime, Japan, 791-0295 Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 812-8582 Novartis Investigative Site Kurume-city, Fukuoka, Japan, 830-0011 Novartis Investigative Site Maebashi-city, Gunma, Japan, 371-8511 Novartis Investigative Site Sapporo-city, Hokkaido, Japan, 060-8543 Novartis Investigative Site Sapporo-city, Hokkaido, Japan, 060-8648 Novartis Investigative Site Kobe-city, Hyogo, Japan, 650-0017 Novartis Investigative Site Kobe-city, Hyogo, Japan, 650-0047 Novartis Investigative Site Kumamoto City, Kumamoto, Japan, 860-8556 Novartis Investigative Site Kyoto-city, Kyoto, Japan, 606-8507 Novartis Investigative Site Tsu-city, Mie, Japan, 514-8507 Novartis Investigative Site Sendai-city, Miyagi, Japan, 980-8574 Novartis Investigative Site Miyazaki-city, Miyazaki, Japan, 889-1692 Novartis Investigative Site Okayama-city, Okayama, Japan, 700-8558 Novartis Investigative Site Hirakata-city, Osaka, Japan, 573-1191 Novartis Investigative Site OsakaSayama, Osaka, Japan, 589-8511 Novartis Investigative Site Suita-city, Osaka, Japan, 565-0871 Novartis Investigative Site Shimotsuke-city, Tochigi, Japan, 329-0498 Novartis Investigative Site Bunkyo-ku, Tokyo, Japan, 113-8431 Novartis Investigative Site Bunkyo-ku, Tokyo, Japan, 113-8519 Novartis Investigative Site Bunkyo-ku, Tokyo, Japan, 113-8603 Novartis Investigative Site Bunkyo-ku, Tokyo, Japan, 113-8677 Novartis Investigative Site Shinjuku-ku, Tokyo, Japan, 160-0023 Novartis Investigative Site Shinjuku-ku, Tokyo, Japan, 162-8666 Novartis Investigative Site Chuo-city, Yamanashi, Japan, 409-3898 Novartis Investigative Site Akita, Japan, 010-8543 Novartis Investigative Site Gifu, Japan, 501-1194 Collapse << |
| NCT02087059 | - | - | Completed | - | - |
| NCT03373877 | Myelofibrosis ... more >> Primary Myelofibrosis Post-polycythemia Vera Myelofibrosis Post-essential Thrombocythemia Myelofibrosis Collapse << | Phase 1 | Recruiting | December 2019 | United States, Connecticut ... more >> Yale Cancer Center Recruiting New Haven, Connecticut, United States, 06511 Contact: Kris Awerkamp, BSN RN CCRP 602-358-8319 kawerkamp@td2inc.com Principal Investigator: Nikolai Podoltsev, MD PhD United States, Louisiana Ochsner Clinic Foundation Recruiting New Orleans, Louisiana, United States, 70121 Contact: Kris Awerkamp, BSN, RN 602-358-8319 kawerkamp@td2inc.com Principal Investigator: Laura Finn, MD United States, Michigan University of Michigan Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Kris Awerkamp, BSN, RN 602-358-8319 kawerkamp@td2inc.com Principal Investigator: Moshe Talpaz, MD Karmanos Cancer Institute Recruiting Detroit, Michigan, United States, 48201 Contact: Kris Awerkamp, BSN, RN 602-358-8319 kawerkamp@td2inc.com Principal Investigator: Jay Yang, MD United States, Nebraska University of Nebraska Medical Center Recruiting Omaha, Nebraska, United States, 68198 Contact: Kris Awerkamp, BSN, RN 602-358-8319 kawerkamp@td2inc.com Principal Investigator: Krishna Gundabolu, MD United States, North Carolina Duke University Medical Center Recruiting Durham, North Carolina, United States, 27710 Contact: Kris Awerkamp, BSN RN CCRP 602-358-8319 kawerkamp@td2inc.com Principal Investigator: Murat Arcasoy, MD United States, Ohio Cleveland Clinic - Taussig Cancer Institute Recruiting Cleveland, Ohio, United States, 44195 Contact: Kris Awerkamp, BSN RN CCRP 602-358-8319 kawerkamp@td2inc.com Principal Investigator: Moshe Talpaz, MD United States, Pennsylvania Abramson Cancer Center - University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Kris Awerkamp, BSN, RN 602-358-8319 kawerkamp@td2inc.com Principal Investigator: Elizabeth Hexner, MD United States, Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Kris Awerkamp, BSN, RN 602-358-8319 kawerkamp@td2inc.com Principal Investigator: Naveen Pemmaraju, MD Mays Cancer Center UT Health San Antonio Recruiting San Antonio, Texas, United States, 78229-3900 Contact: Kris Awerkamp, BSN, RN, CCRP 602-358-8319 kawerkamp@td2inc.com Principal Investigator: Ruben A. Mesa, MD, FACP Collapse << |
| NCT02913261 | Corticosteroid Refractory Acut... more >>e Graft vs Host Disease Collapse << | Phase 3 | Recruiting | March 8, 2021 | - |
| NCT00639002 | Multiple Myeloma | Phase 2 | Completed | - | United States, California ... more >> Highland, California, United States, 92346 United States, Florida Boynton Beach, Florida, United States, 33435 United States, New York New York, New York, United States, 10011 Collapse << |
| NCT02475655 | HIV Infections | Phase 2 | Completed | - | United States, Alabama ... more >> Alabama CRS Birmingham, Alabama, United States, 35294 United States, California UCLA CARE Center CRS Los Angeles, California, United States, 90035 UCSD Antiviral Research Center CRS San Diego, California, United States, 92103 Ucsf Hiv/Aids Crs San Francisco, California, United States, 94110 United States, Illinois Northwestern University CRS Chicago, Illinois, United States, 60611 United States, Missouri Washington University Therapeutics (WT) CRS Saint Louis, Missouri, United States, 63110-1010 United States, New York Weill Cornell Chelsea CRS New York, New York, United States, 10010 Weill Cornell Uptown CRS New York, New York, United States, 10065 University of Rochester Adult HIV Therapeutic Strategies Network CRS Rochester, New York, United States, 14642 United States, Ohio Cincinnati Clinical Research Site Cincinnati, Ohio, United States, 45219 Case Clinical Research Site Cleveland, Ohio, United States, 44106 United States, Pennsylvania Penn Therapeutics, CRS Philadelphia, Pennsylvania, United States, 19104 United States, Rhode Island The Miriam Hospital Clinical Research Site (TMH CRS) CRS Providence, Rhode Island, United States, 02906 United States, Tennessee Vanderbilt Therapeutics (VT) CRS Nashville, Tennessee, United States, 37204 Collapse << |
| NCT00509899 | - | - | Completed | - | - |
| NCT02119676 | CRC (Colorectal Cancer) | Phase 2 | Terminated(Substudy 1 was term... more >>inated for futility at interim analysis and Substudy 2 was terminated per sponsor decision.) Collapse << | - | - |
| NCT02119676 | - | - | Terminated(Substudy 1 was term... more >>inated for futility at interim analysis and Substudy 2 was terminated per sponsor decision.) Collapse << | - | - |
| NCT02038036 | Polycythemia Vera | Phase 3 | Active, not recruiting | April 8, 2020 | - |
| NCT01644110 | Primary Myelofibrosis ... more >> Secondary Myelofibrosis PMF SMF Post-PV MF Post-ET MF Collapse << | Phase 1 Phase 2 | Recruiting | May 2022 | Germany ... more >> University of Ulm Recruiting Ulm, Germany, 89081 Contact: Konstanze Doehner, MD konstanze.doehner@uniklinik-ulm.de Principal Investigator: konstanze Doehner, Md Collapse << |
| NCT00509899 | Myelofibrosis ... more >> Polycythemia Vera Thrombocytosis Collapse << | Phase 1 Phase 2 | Completed | - | United States, Minnesota ... more >> Rochester, Minnesota, United States United States, Texas Houston, Texas, United States Collapse << |
| NCT02997280 | Graft Vs Host Disease | Phase 2 | Recruiting | June 2019 | Russian Federation ... more >> First Pavlov State Medical University of St. Petersburg Recruiting Saint-Petersburg, Russian Federation, 197089 Contact: Ivan S. Moiseev, PhD +79217961951 moisiv@mail.ru Contact: Boris V. Afanasyev, Prof +78122334551 coordinatorbmt@gmail.com Collapse << |
| NCT00639002 | - | - | Completed | - | - |
| NCT00726232 | MPN (Myeloproliferative Neopla... more >>sms) Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> Houston, Texas, United States, 77030 Italy Bergamo, Italy Firenze, Italy Pavia, Italy Collapse << |
| NCT02120417 | Breast Cancer | Phase 2 | Terminated(The study was termi... more >>nated as other related studies of ruxolitinib did not provide sufficient efficacy to warrant continuation.) Collapse << | - | - |
| NCT02292446 | Polycythemia Vera | Phase 3 | Completed | - | - |
| NCT01348490 | MPN (Myeloproliferative Neopla... more >>sms) Collapse << | Phase 2 | Active, not recruiting | November 2018 | - |
| NCT02598297 | Early Myelofibrosis With High ... more >>Molecular Risk Mutations Collapse << | Phase 3 | Completed | - | - |
| NCT02257138 | Blasts More Than 20 Percent of... more >> Bone Marrow Nucleated Cells Blasts More Than 20 Percent of Peripheral Blood White Cells Myelodysplastic/Myeloproliferative Neoplasm Recurrent Acute Myeloid Leukemia Refractory Acute Myeloid Leukemia Collapse << | Phase 1 Phase 2 | Recruiting | February 1, 2019 | United States, Texas ... more >> M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Farhad Ravandi-Kashani 713-745-0394 Principal Investigator: Farhad Ravandi-Kashani Collapse << |
| NCT02396628 | Graft vs Host Disease | Phase 2 | Recruiting | March 2023 | Germany ... more >> Charité - Universitätsmedizin Berlin Recruiting Berlin, Germany, 13353 Principal Investigator: Igor Blau, PD Dr. Sub-Investigator: Il-Kang Na, PD Dr. Sub-Investigator: Giang Lam Vuong, Dr. Universitätsklinikum Bonn Recruiting Bonn, Germany, 53105 Principal Investigator: Dominik Wolf, Prof. Sub-Investigator: Karin Mayer, Dr. Universitätsklinikum Dresden Recruiting Dresden, Germany, 01307 Principal Investigator: Friedrich Stölzel, Dr. Sub-Investigator: Rainer Ordemann, Prof. University Medical Center Recruiting Freiburg, Germany, 79106 Contact: Nikolas von Bubnoff, Professor nikolas.bubnoff@uniklinik-freiburg.de Principal Investigator: Nikolas von Bubnoff, Professor Sub-Investigator: Robert Zeiser, Professor Universitätsklinikum Hamburg Eppendorf Recruiting Hamburg, Germany, 20246 Principal Investigator: Francis Ayuk, PD Dr. Sub-Investigator: Dietlinde Janson, Dr. Universitätsklinikum Heidelberg Recruiting Heidelberg, Germany, 69120 Principal Investigator: Peter Dreger, Prof. Sub-Investigator: Thomas Luft, PD Dr. Universitätsklinikum des Saarlandes Recruiting Homburg, Germany, 66421 Contact: Jörg T. Bittenbring, Dr. Principal Investigator: Jörg T. Bittenbring, Dr. Sub-Investigator: Niels Murawski, PD Dr. Universitätsklinikum Köln Recruiting Köln, Germany, 50937 Contact: Udo Holtick, Dr. Principal Investigator: Udo Holtick, Dr. Sub-Investigator: Christof Scheid, Prof. Sub-Investigator: Marco Herling, Dr. Universitätsklinikum Marburg Recruiting Marburg, Germany, 35043 Contact: Andreas Burchert, Prof. Principal Investigator: Andreas Burchert, Prof. Sub-Investigator: Kristina Sohlbach, Dr. Universitätsklinikum München TU rechts der Isar Recruiting München, Germany, 81675 Principal Investigator: Mareike Verbeek, Dr. Sub-Investigator: Sandra Grass, Dr. Universitätsklinikum Würzburg Recruiting Würzburg, Germany, 97080 Principal Investigator: Götz Grigoleit, Dr. Sub-Investigator: Sabrina Kraus, Dr. Collapse << |
| NCT03144687 | MPN (Myeloproliferative Neopla... more >>sms) Collapse << | Phase 2 | Recruiting | March 2019 | United States, Arizona ... more >> Arizona Oncology Associates Recruiting Tempe, Arizona, United States, 85284 Contact: Study Coordinator 480-256-1664 Principal Investigator: Kasra Karamlou United States, California UC Irvine Medical Center Recruiting Orange, California, United States, 92868 Contact: Study Coordinator 714-456-5408 Principal Investigator: Angela Fleischman, MD United States, Colorado Anschutz Cancer Pavilion - University Of Colorado Recruiting Aurora, Colorado, United States, 80045 Contact: Study Coordinator 720-848-2587 Principal Investigator: Brandon McMahon United States, Connecticut Norwalk Hospital Recruiting Norwalk, Connecticut, United States, 06856 Contact: Study Coordinator 203-852-2996 Principal Investigator: Richard Frank United States, District of Columbia Georgetown University Hospital Not yet recruiting Washington, District of Columbia, United States, 20007 Contact: Study Coordinator 202-687-0116 Principal Investigator: Craig Kessler United States, Indiana Parkview Research Center Recruiting Fort Wayne, Indiana, United States, 46845 Contact: Study Coordinator 260-425-6831 Principal Investigator: Robert Manges United States, Maryland University Of Maryland - Greenebaum Cancer Center Recruiting Baltimore, Maryland, United States, 21201 Contact: Study Coordinator 410-328-6635 Principal Investigator: Duong United States, Michigan University of Michigan Health System Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Study Coordinator 734-936-3879 Principal Investigator: Moshe Talpaz, MD Karmanos Cancer Institute Recruiting Detroit, Michigan, United States, 48201 Contact: Study Coordinator 313-576-9758 Principal Investigator: Jay Yang Providence Cancer Center Recruiting Southfield, Michigan, United States, 48075 Contact: Study Coordinator 248-849-5332 Principal Investigator: Howard Terebelo United States, New Mexico University Of New Mexico Cancer Center Recruiting Albuquerque, New Mexico, United States, 87102 Contact: Study Coordinator 505-925-0379 Principal Investigator: Arana Yi United States, New York Weill Cornell Medical Centers Not yet recruiting New York, New York, United States, 10065 Contact: Study Coordinator 212-746-2389 Principal Investigator: Ellen Ritchie New York Medical College Not yet recruiting Valhalla, New York, United States, 10595 Contact: Study Coordinator 914-493-8375 Principal Investigator: Karen Seiter United States, North Carolina Duke University Medical Center Not yet recruiting Durham, North Carolina, United States, 27710 Contact: Study Coordinator 919-681-4769 Principal Investigator: Murat Arcasoy United States, Ohio Cleveland Clinic Recruiting Cleveland, Ohio, United States, 44195 Contact: Study Coordinator 216-444-9464 Principal Investigator: Aaron Gerds, MD United States, Oregon Willamette Valley Cancer Institute Recruiting Eugene, Oregon, United States, 97401 Contact: Study Coordinator 281-863-4631 Principal Investigator: Jeff Sharman, MD United States, Texas Texas Oncology San Antonio Recruiting San Antonio, Texas, United States, 78240 Contact: Study Coordinator 210-595-5680 Principal Investigator: Roger Lyons Texas Oncology - Tyler Recruiting Tyler, Texas, United States, 75702 Contact: Study Coordinator 903-579-9800 Principal Investigator: Habte Yimer United States, Virginia University of Virginia Recruiting Charlottesville, Virginia, United States, 22908 Contact: Study Coordinator 434-982-3927 Principal Investigator: Michael Keng, MD Collapse << |
| NCT00638378 | Prostate Cancer | Phase 2 | Terminated(According to the pr... more >>otocol, the sponsor terminated the study after it was determined that less than 2 of the first 22 patients showed a PSA50 response.) Collapse << | - | United States, California ... more >> Highland, California, United States Montebello, California, United States Mountain View, California, United States United States, Illinois Galesburg, Illinois, United States United States, Kansas Overland Park, Kansas, United States Wichita, Kansas, United States United States, Michigan Grand Rapids, Michigan, United States United States, Missouri Jefferson City, Missouri, United States United States, Montana Great Falls, Montana, United States, 59405 United States, New Jersey Cherry Hill, New Jersey, United States United States, New York Staten Island, New York, United States United States, North Dakota Bismarck, North Dakota, United States United States, Pennsylvania Bethlehem, Pennsylvania, United States United States, South Carolina Sumter, South Carolina, United States United States, Washington Lacey, Washington, United States Collapse << |
| NCT02131584 | Chronic Lymphocytic Leukemia ... more >> Fatigue Collapse << | Phase 2 | Active, not recruiting | September 30, 2022 | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01431209 | Recurrent Diffuse Large B-Cell... more >> Lymphoma Recurrent Mature T- and NK-Cell Non-Hodgkin Lymphoma Refractory Diffuse Large B-Cell Lymphoma Refractory Mature T-Cell and NK-Cell Non-Hodgkin Lymphoma Collapse << | Phase 2 | Active, not recruiting | December 2019 | United States, Maryland ... more >> National Institutes of Health Clinical Center Bethesda, Maryland, United States, 20892 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States, 68198 Collapse << |
| NCT02436135 | Myelofibrosis | Phase 1 | Completed | - | United States, California ... more >> Stanford Hospital and Clinics Stanford, California, United States, 94305 United States, Michigan University of Michigan Health System Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT02955940 | Pancreatic Cancer ... more >> Colorectal Cancer (CRC) Breast Cancer Lung Cancer Collapse << | Phase 2 | Enrolling by invitation | November 2019 | United States, Kentucky ... more >> University of Louisville Louisville, Kentucky, United States, 40202 United States, New York New York Oncology Hematology PC Hudson, New York, United States, 12534 United States, Texas U of Texas M.D. Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02076191 | Myeloproliferative Neoplasms | Phase 1 Phase 2 | Active, not recruiting | December 2018 | United States, Missouri ... more >> Washington University of St. Louis Saint Louis, Missouri, United States, 63110 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Columbia University Medical Center New York, New York, United States, 10032 Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 United States, North Carolina Wake Forest University Baptist Medical Center Winston-Salem, North Carolina, United States, 27157 United States, Ohio Cleveland Clinic Taussig Cancer Center Institute Cleveland, Ohio, United States, 44195 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00638378 | - | - | Terminated(According to the pr... more >>otocol, the sponsor terminated the study after it was determined that less than 2 of the first 22 patients showed a PSA50 response.) Collapse << | - | - |
| NCT03701698 | Acute GVHD | Phase 2 | Not yet recruiting | October 31, 2021 | China, Shanghai ... more >> Shanghai General Hospital Active, not recruiting Shanghai, Shanghai, China, 200080 Collapse << |
| NCT01562873 | Breast Cancer | Phase 2 | Terminated(Not enough response... more >>s to continue treatment.) Collapse << | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01776723 | Myelomonocytic Leukemia | Phase 1 Phase 2 | Active, not recruiting | April 2020 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 United States, Maryland Johns Hopkins Sidney Kimmel Comprehensive Cancer Center Baltimore, Maryland, United States, 21287 United States, Massachusetts Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, New York Weill Medical College of Cornell New York, New York, United States, 10021 United States, Ohio Cleveland Clinic Cleveland, Ohio, United States, 44106 Collapse << |
| NCT01562873 | - | - | Terminated(Not enough response... more >>s to continue treatment.) Collapse << | - | - |
| NCT02400463 | Hemophagocytic Syndrome (HPS) | Early Phase 1 | Recruiting | November 2018 | United States, Michigan ... more >> University of Michigan Comprehensive Cancer Center Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Ryan Wilcox, M.D. 734-615-1482 rywilcox@umich.edu Principal Investigator: Ryan Wilcox, M.D. Collapse << |
| NCT02966353 | Primary Myelofibrosis ... more >> Post-Polycythemia Vera-Myelofibrosis Post-Essential Thrombocythemia Myelofibrosis Collapse << | Phase 2 | Active, not recruiting | February 15, 2019 | Austria ... more >> Novartis Investigative Site Vienna, Austria, A-1090 Belgium Novartis Investigative Site Antwerpen, Belgium, 2060 Novartis Investigative Site Leuven, Belgium, 3000 Bulgaria Novartis Investigative Site Sofia, Bulgaria, 1413 Novartis Investigative Site Sofia, Bulgaria, 1756 Canada, British Columbia Novartis Investigative Site Vancouver, British Columbia, Canada, V5Z 1M9 Germany Novartis Investigative Site Halle/S., Germany, 06120 Greece Novartis Investigative Site Thessaloniki, GR, Greece, 570 10 Novartis Investigative Site Athens, Greece, 11527 Italy Novartis Investigative Site Bologna, BO, Italy, 40138 Novartis Investigative Site Firenze, FI, Italy, 50134 Novartis Investigative Site Palermo, PA, Italy, 90127 Japan Novartis Investigative Site Bunkyo-ku, Tokyo, Japan, 113-8431 Russian Federation Novartis Investigative Site Moscow, Russian Federation, 125167 Novartis Investigative Site Moscow, Russian Federation, 129110 Novartis Investigative Site Petrozavodsk, Russian Federation, 185019 Spain Novartis Investigative Site Barcelona, Catalunya, Spain, 08036 Novartis Investigative Site Alicante, Comunidad Valenciana, Spain, 03010 Novartis Investigative Site Santiago de Compostela, Galicia, Spain, 15706 Turkey Novartis Investigative Site Istanbul, Turkey, 34093 Novartis Investigative Site Kocaeli, Turkey, 41380 Collapse << |
| NCT02091752 | Primary Myelofibrosis | Phase 2 | Terminated(The study was termi... more >>nated due to low enrollment.) Collapse << | - | Germany ... more >> Novartis Investigative Site Leipzig, Germany, 04103 Italy Novartis Investigative Site Firenze, FI, Italy, 50134 Spain Novartis Investigative Site Madrid, Spain, 28034 Collapse << |
| NCT02253277 | Chronic Myeloid Leukemia | Phase 1 Phase 2 | Completed | - | Germany ... more >> Novartis Investigative Site Berlin, Germany, 13353 Novartis Investigative Site Frankfurt, Germany, 60590 Novartis Investigative Site Freiburg, Germany, 79106 Novartis Investigative Site Jena, Germany, 07740 Novartis Investigative Site Leipzig, Germany, 04103 Novartis Investigative Site Mannheim, Germany, 68169 Collapse << |
| NCT02091752 | - | - | Terminated(The study was termi... more >>nated due to low enrollment.) Collapse << | - | - |
| NCT01251965 | Leukemia | Phase 1 Phase 2 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00726232 | - | - | Completed | - | - |
| NCT01702064 | Chronic Phase Chronic Myeloid ... more >>Leukemia Collapse << | Phase 1 | Active, not recruiting | March 2019 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT02876302 | Inflammatory Breast Cancer (IB... more >>C) Collapse << | Phase 2 | Recruiting | February 2024 | United States, Maryland ... more >> Johns Hopkins University Recruiting Baltimore, Maryland, United States, 21287 Contact: Vered Stearns, MD vstearn1@jhmi.edu Johns Hopkins at Green Spring Station Recruiting Lutherville, Maryland, United States, 21093 Contact: Vered Stearns, MD Vstearn1@jhmi.edu United States, Massachusetts Dana-Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 Contact: Beth Overmoyer, MD, FACP 617-632-4056 bovermoyer@partners.org Principal Investigator: Beth Overmoyer, MD, FACP United States, Michigan University of Michigan Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Catherine Van Poznak, MD cvanpoz@med.umich.edu United States, Minnesota Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Tufia Haddad, MD Haddad.Tufia@mayo.edu United States, North Carolina Duke University Medical Center Recruiting Durham, North Carolina, United States, 27710 Contact: Jeremy Force, MD Jeremy.force@duke.edu United States, Pennsylvania University of Pennslyvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Amy Clark, MD Clarkamy@mail.upenn.edu United States, Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Naoto Ueno, MD Nueno@mdanderson.org Collapse << |
| NCT01340651 | - | - | Completed | - | - |
| NCT01340651 | Myelofibrosis | Phase 2 | Completed | - | United States, Arizona ... more >> Scottsdale, Arizona, United States United States, Florida Winter Park, Florida, United States United States, Texas Houston, Texas, United States, TX Collapse << |
| NCT02155465 | Lung Cancer | Phase 1 Phase 2 | Completed | - | United States, New Jersey ... more >> Memoral Sloan Kettering Cancer Center Basking Ridge, New Jersey, United States United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering West Harrison Harrison, New York, United States, 10604 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan Kettering at Mercy Medical Center Rockville Centre, New York, United States Memoral Sloan Kettering Cancer Center at Phelps Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT01445769 | Primary Myelofibrosis ... more >> Post-Polycythemia Vera Myelofibrosis Post-Essential Thrombocythemia Myelofibrosis Collapse << | Phase 2 | Completed | - | United States, California ... more >> Highland, California, United States La Jolla, California, United States Los Angeles, California, United States United States, Florida Jacksonville, Florida, United States Orange City, Florida, United States Winter Park, Florida, United States United States, Georgia Atlanta, Georgia, United States Augusta, Georgia, United States United States, Iowa Iowa City, Iowa, United States United States, Maryland Baltimore, Maryland, United States United States, Michigan Ann Arbor, Michigan, United States Southfield, Michigan, United States United States, New Jersey Morristown, New Jersey, United States United States, New York Armonk, New York, United States United States, North Carolina Hickory, North Carolina, United States United States, Ohio Canton, Ohio, United States United States, Pennsylvania Hazelton, Pennsylvania, United States Hershey, Pennsylvania, United States United States, South Carolina Charleston, South Carolina, United States United States, South Dakota Sioux Falls, South Dakota, United States United States, Texas San Antonio, Texas, United States Collapse << |
| NCT01445769 | - | - | Completed | - | - |
| NCT03610971 | Chronic Phase Chronic Myeloid ... more >>Leukemia Chronic Myeloid Leukemia, Chronic Phase Collapse << | Phase 2 | Not yet recruiting | January 2022 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Not yet recruiting Tampa, Florida, United States, 33612 Contact: Anthony McLaughlin 813-745-5941 anthony.mclaughlin@moffitt.org Contact: Kendra Sweet, M.D. 813-745-8986 kendra.sweet@moffitt.org Principal Investigator: Kendra Sweet, M.D. Collapse << |
| NCT01693601 | Myelofibrosis | Phase 1 Phase 2 | Active, not recruiting | February 2019 | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT02015208 | Chronic Lymphocytic Leukemia | Phase 1 Phase 2 | Completed | - | Canada, Ontario ... more >> Sunnybrook Odette Cancer Center Toronto, Ontario, Canada, M4N3M5 Collapse << |
| NCT03571321 | Acute Lymphoblastic Leukemia ... more >> ALL, Childhood ALL Collapse << | Phase 1 | Not yet recruiting | September 5, 2024 | United States, Illinois ... more >> University of Chicago Medical Center Not yet recruiting Chicago, Illinois, United States, 60637 Contact: Howie Weiner 773-702-2084 hweiner@medicine.bsd.uchicago.edu Contact: Peggy Green, RN (773) 702-0267 mgreen@medicine.bsd.uchicago.edu Principal Investigator: Emily Currant, MD Collapse << |
| NCT03069326 | Myelofibrosis | Phase 2 | Recruiting | February 2019 | United States, New Jersey ... more >> Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Raajit Rampal, MD, PhD 212-639-2194 United States, New York Memorial Sloan Kettering Commack Recruiting Commack, New York, United States, 11725 Contact: Raajit Rampa, MD, PhD 212-639-2194 Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Raajit Rampal, MD, PhD 212-639-2194 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Raajit Rampal, MD, PhD 212-639-2194 Contact: Ellin Berman, MD 212-639-7762 Principal Investigator: Raajit Rampal, MD, PhD Memorial Sloan Kettering Rockville Centre Recruiting Rockville Centre, New York, United States, 11570 Contact: Raajit Rampal, MD, PhD 212-639-2194 United States, Texas Md Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Prithvira Bose, MD 713-792-7747 Principal Investigator: Prithvira Bose, MD Collapse << |
| NCT01950780 | Alopecia Areata | Phase 2 | Completed | - | United States, New York ... more >> Columbia University Medical Center, Department of Dermatology New York, New York, United States, 10032 Collapse << |
| NCT02613598 | Hodgkin's Lymphoma ... more >> Lymphoma, Non-Hodgkin Collapse << | Phase 1 | Recruiting | October 2021 | United States, Michigan ... more >> University of Michgan Comprehensive Cancer Center Recruiting Ann Arbor, Michigan, United States, 48187 Contact: Tycel Phillips, M.D. 734-232-2883 tycelp@umich.edu Principal Investigator: Tycel Phillips, M.D. Collapse << |
| NCT02066532 | Metastatic Breast Cancer ... more >> Breast Carcinoma HER-2 Positive Breast Cancer Collapse << | Phase 1 Phase 2 | Recruiting | December 2018 | United States, New York ... more >> Mount Sinai Medical Center Recruiting New York, New York, United States, 10029 Contact: Amy Tiersten, MD 212-241-2299 amy.tiersten@mssm.edu Principal Investigator: Amy Tiersten, MD Columbia University Medical Center Recruiting New York, New York, United States, 10032 Contact: Kevin Kalinsky, MD, MS 212-305-1945 KK2693@cumc.columbia.edu Contact: Dan Otap, CCRP 212-342-3970 DO2267@cumc.columbia.edu Principal Investigator: Kevin Kalinsky, MD, MS New York Hospital-Weill Cornell Medical Center Recruiting New York, New York, United States, 10065 Contact: Tessa Cigler, MD 212-821-0644 elc2007@med.cornell.edu Principal Investigator: Tessa Cigler, MD Montefiore Medical Center Recruiting New York, New York, United States, 10461 Contact: Della Makower, MD 718-405-8529 dmakower@montefiore.org Principal Investigator: Della Makower, MD Collapse << |
| NCT03674047 | Other Cancer | Phase 2 | Not yet recruiting | March 31, 2023 | United States, Massachusetts ... more >> Dana Farber Cancer Institute Not yet recruiting Boston, Massachusetts, United States, 02214 Contact: Zachariah DeFilipp, MD 617-724-4000 zdefilipp@mgh.harvard.edu Principal Investigator: Zachariah DeFilipp, MD Collapse << |
| NCT03491215 | Acute Graft Versus Host Diseas... more >>e Collapse << | Phase 1 Phase 2 | Recruiting | July 1, 2022 | Australia, Queensland ... more >> Novartis Investigative Site Recruiting Brisbane, Queensland, Australia, 4101 Australia, Victoria Novartis Investigative Site Recruiting Parkville, Victoria, Australia, 3052 Belgium Novartis Investigative Site Recruiting Bruxelles, Belgium, 1200 Novartis Investigative Site Recruiting Leuven, Belgium, 3000 Italy Novartis Investigative Site Recruiting Genova, GE, Italy, 16147 Slovenia Novartis Investigative Site Recruiting Ljubljana, Slovenia, 1000 Spain Novartis Investigative Site Recruiting Barcelona, Cataluña, Spain, 08025 Novartis Investigative Site Recruiting Madrid, Spain, 28009 Collapse << |
| NCT03222609 | Myelofibrosis (MF) | Phase 2 | Recruiting | June 11, 2021 | United States, Alabama ... more >> UAB Comprehensive Cancer Cente /ID# 165464 Recruiting Birmingham, Alabama, United States, 35217 United States, California Ucsd /Id# 164084 Recruiting La Jolla, California, United States, 92037 Usc /Id# 164095 Recruiting Los Angeles, California, United States, 90033 United States, Florida Mayo Clinic /ID# 164201 Recruiting Jacksonville, Florida, United States, 32224 Moffitt Cancer Center /ID# 164082 Recruiting Tampa, Florida, United States, 33612 United States, Illinois University of Chicago /ID# 164115 Recruiting Chicago, Illinois, United States, 60637 United States, Indiana Indiana Blood & Marrow Transpl /ID# 165075 Recruiting Indianapolis, Indiana, United States, 46237 United States, Massachusetts Dana-Farber Cancer Institute /ID# 162675 Recruiting Boston, Massachusetts, United States, 02215 United States, Michigan Henry Ford Hospital /ID# 162682 Recruiting Detroit, Michigan, United States, 48202 United States, New York Weill Cornell Medical College /ID# 162679 Recruiting New York, New York, United States, 10021 United States, Texas MD Anderson Cancer Center /ID# 162683 Recruiting Houston, Texas, United States, 77030 UT Health Cancer Center /ID# 164094 Recruiting San Antonio, Texas, United States, 78229 United States, Utah University of Utah /ID# 164116 Recruiting Salt Lake City, Utah, United States, 84112-5500 United Kingdom Guy's and St Thomas' NHS Found /ID# 164110 Recruiting London, London, City Of, United Kingdom, SE1 9RT Christie NHS Foundation Trust /ID# 164111 Recruiting Manchester, United Kingdom, M20 4BX Collapse << |
| NCT02469974 | Myelofibrosis ... more >> MF Collapse << | Not Applicable | Withdrawn(no enrollments) | - | - |
| NCT03613428 | Acute T Cell Leukemia | Phase 1 Phase 2 | Not yet recruiting | March 30, 2021 | - |
| NCT02723994 | Leukemia | Phase 2 | Recruiting | May 2024 | - |
| NCT03745651 | Atopic Dermatitis | Phase 3 | Recruiting | September 2020 | United States, Florida ... more >> San Marcus Research Clinic Inc Recruiting Miami, Florida, United States, 33015 Well Pharma Medical Research Corporation Not yet recruiting Miami, Florida, United States, 33143 Forward Clinical Trials Inc Not yet recruiting Tampa, Florida, United States, 33624 United States, Georgia Clinical Research Atlanta Not yet recruiting Stockbridge, Georgia, United States, 30281 United States, Kentucky Derm Research LLC Not yet recruiting Louisville, Kentucky, United States, 40217 Dermatology Specialists PSC Not yet recruiting Louisville, Kentucky, United States, 40241 United States, New Hampshire ActivMed Practices & Research Inc Not yet recruiting Portsmouth, New Hampshire, United States, 03801 United States, New Jersey Hassman Research Institute Not yet recruiting Berlin, New Jersey, United States, 08009 United States, Ohio Synexus Clinical Research US, Inc. - Cincinnati Not yet recruiting Cincinnati, Ohio, United States, 45236 United States, Pennsylvania Paddington Testing Company Inc Not yet recruiting Philadelphia, Pennsylvania, United States, 19103 United States, South Carolina Synexus Clinical Research US, Inc. - Anderson Not yet recruiting Anderson, South Carolina, United States, 29621 United States, Texas Clinical Trials of Texas Incorporated Not yet recruiting San Antonio, Texas, United States, 78229 Center for Clinical Studies Not yet recruiting Webster, Texas, United States, 77598 United States, Virginia Clinical Research Partners LLC Not yet recruiting Henrico, Virginia, United States, 23233 United States, Washington Dermatology Specialists of Spokane Not yet recruiting Spokane, Washington, United States, 99204 Collapse << |
| NCT03112603 | Graft-versus-host Disease (GVH... more >>D) Collapse << | Phase 3 | Recruiting | January 2022 | - |
| NCT03153982 | Head and Neck Squamous Cell Ca... more >>rcinoma Collapse << | Phase 2 | Not yet recruiting | September 29, 2020 | United States, Arizona ... more >> University of Arizona Cancer Center Not yet recruiting Tucson, Arizona, United States, 85724 Contact: Clinical Trials Office 866-278-1554 UACC-C3@uacc.arizona.edu Sub-Investigator: Julie Bauman, MD United States, California UCSF Helen Diller Family Comprehensive Cancer Center Not yet recruiting San Francisco, California, United States, 94131 Contact: Kyusun Cha, CCRC 415-502-3081 Kyusun.Cha@ucsf.edu Collapse << |
| NCT02973711 | Leukemia, Chronic Myeloid | Phase 1 Phase 2 | Recruiting | April 2021 | United States, Michigan ... more >> The University of Michigan Comprehensive Cancer Center Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Patrick Burke, M.D. 734-647-8921 pwburke@umich.edu Principal Investigator: Patrick Burke, M.D. United States, North Carolina Wake Forest University Health Sciences Not yet recruiting Winston-Salem, North Carolina, United States, 27157 Contact: Rupali Bhave, M.D. 336-713-0864 rbhave@wakehealth.edu Contact: Megan Brown-Farmer 336-713-6913 mebrown@wakehealth.edu Collapse << |
| NCT02953678 | Graft-versus-host Disease (GVH... more >>D) Collapse << | Phase 2 | Active, not recruiting | June 2020 | - |
| NCT03745638 | Atopic Dermatitis | Phase 3 | Recruiting | September 2020 | - |
| NCT03147742 | - | - | - | - | United States, Florida ... more >> Blood & Marrow Transplant Center Orlando, Florida, United States, 32804 Principal Investigator: Dr. Steven Goldstein United States, Kansas Cancer Center of Kansas Wichita, Kansas, United States, 67214 Principal Investigator: Dr. Shaker Dakhil United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02215 Principal Investigator: Dr. Yin-Bin Chen United States, Michigan University of Michigan Comprehensive Cancer Center Ann Arbor, Michigan, United States, 48109-5271 Principal Investigator: Dr. Sarah Anand United States, New Jersey Hackensack University Medical Center Hackensack, New Jersey, United States, 07601 Principal Investigator: Dr. Alfred Gillio United States, New York Weill Cornell Medical College New York, New York, United States, 10021 Principal Investigator: Dr. Tsiporah Shore Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Principal Investigator: Dr. Miguel-Angel Perales United States, Oregon Oregon Health & Science University Portland, Oregon, United States, 97239 Principal Investigator: Dr. Rachel Cook United States, Pennsylvania Gettysburg Cancer Center Gettysburg, Pennsylvania, United States, 17325 Principal Investigator: Dr. Satish Shah Allegheny Health Network Cancer Institute Pittsburgh, Pennsylvania, United States, 15224 Principal Investigator: Dr. Anna Koget United States, South Carolina Greenville Health System Cancer Institute Greenville, South Carolina, United States, 29615 Principal Investigator: Dr. Suzanne Fanning United States, South Dakota Avera Research Institute Sioux Falls, South Dakota, United States, 57105 Principal Investigator: Dr. Vinod Parameswaran United States, Texas Medical City Dallas Hospital Dallas, Texas, United States, 75230 Principal Investigator: Dr. Vikas Bhushan United States, Washington Fred Hutchinson Cancer Research Center Seattle, Washington, United States, 98109-1024 Principal Investigator: Dr. Paul Carpenter United States, Wisconsin Medical College of Wisconsin Milwaukee, Wisconsin, United States, 53226 Principal Investigator: Dr. Parameswaran Hari Collapse << |
| NCT03110822 | Multiple Myeloma | Phase 1 | Recruiting | May 2020 | United States, California ... more >> Comprehensive Blood and Cancer Center Recruiting Bakersfield, California, United States, 93309-0633 Contact: Nicole Henry 661-862-8548 nhenry@cbccusa.com Principal Investigator: Ravindranath Patel, MD California Cancer Associates for Research & Excellence (cCARE) Recruiting Encinitas, California, United States, 92024 Contact: Daniela Slavin, PhD 760-452-3909 dslavin@ccare.com Principal Investigator: Alberto Bessudo, MD Robert A. Moss, M.D., F.A.C.P., Inc. Recruiting Fountain Valley, California, United States, 92708 Contact: Helen Tam 714-641-1128 helen.tam@sbcglobal.net Principal Investigator: Robert A Moss, MD Pacific Cancer Care Recruiting Monterey, California, United States, 93940 Contact: Dorothy Tenney 831-375-4105 dtenney@pacificcancercare.com Principal Investigator: Laura Stampleman, MD Sansum Clinic- Ridley-Tree Cancer Center Recruiting Santa Barbara, California, United States, 93105 Contact: Heidi Heitkamp, PhD 805-879-5091 hheitkam@ridleytreecc.org Principal Investigator: Daniel R Greenwald, MD James R. Berenson M.D. Inc. Recruiting West Hollywood, California, United States, 90069 Contact: Regina Swift, R.N 310-623-1227 rswift@berensononcology.com Principal Investigator: James R Berenson, MD United States, Florida Millennium Oncology Research Clinic Recruiting Pembroke Pines, Florida, United States, 33024 Contact: Maria Contreras, MD 954-266-7885 mcontreras@millenniumoncologyfl.com Principal Investigator: Isaac Levy, MD United States, Maryland Regional Cancer Care Associates (RCCA) MD, LLC Recruiting Bethesda, Maryland, United States, 20817 Contact: Natalie Bongiorno, RN 301-571-2016 nbongiorno@regionalcancercare.org Principal Investigator: Ralph Boccia, MD Collapse << |
| NCT02593929 | Head and Neck Squamous Cell Ca... more >>rcinoma Collapse << | Early Phase 1 | Withdrawn | - | - |
| NCT01790295 | Primary Myelofibrosis ... more >> Post Polycythemia Vera Myelofibrosis Post Essential Thrombocythemia Myelofibrosis Collapse << | Phase 2 | Active, not recruiting | February 2021 | United States, Georgia ... more >> Emory Hospital Atlanta, Georgia, United States, 30322 United States, Illinois Northwestern University, Robert h. Lurie Comprehensive Cancer Center Chicago, Illinois, United States, 60611 United States, Kansas University of Kansas Cancer Center Westwood, Kansas, United States, 66205 United States, New York Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 United States, North Carolina Wake Forest Baptist Medical Center Winston-Salem, North Carolina, United States, 27103 United States, Ohio Ohio State University Columbus, Ohio, United States, 43210 Canada Princess Margaret Cancer Centre, University of Toronto Toronto, Canada, M5G 2M9 United Kingdom University of Oxford Oxford, United Kingdom, OX3 9DS Collapse << |
| NCT02267278 | Myeloproliferative Diseases | Phase 2 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT03616184 | Graft-versus-host-disease (GVH... more >>D) Collapse << | Phase 2 | Recruiting | April 2024 | United States, Nebraska ... more >> University of Nebraska Medical Center Recruiting Omaha, Nebraska, United States, 68198 Contact: Penny Nurse Coordinator, RN 402-559-4596 Collapse << |
| NCT03681561 | Hodgkin Lymphoma | Phase 1 Phase 2 | Recruiting | March 2024 | United States, Minnesota ... more >> University of Minnesota Recruiting Minneapolis, Minnesota, United States, 55455 Contact: Erin Zielinski 612-624-0937 eezielin@umn.edu Principal Investigator: Veronika Bachanova, MD Collapse << |
| NCT01895842 | Leukemia | Phase 1 | Active, not recruiting | February 2021 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT03123588 | MPN (Myeloproliferative Neopla... more >>sms) Collapse << | Phase 2 | Recruiting | June 2020 | - |
| NCT03514069 | Glioma Gliobl... more >>astoma Collapse << | Phase 1 | Recruiting | December 2020 | United States, Ohio ... more >> Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Recruiting Cleveland, Ohio, United States, 44195 Contact: Manmeet Ahluwalia, MD 866-223-8100 CancerCenterResearch@ccf.org Principal Investigator: Manmeet Ahluwalia, MD Collapse << |
| NCT02493530 | Myelofibrosis ... more >> Polycythemia Vera Collapse << | Phase 1 | Recruiting | December 2019 | United States, Arizona ... more >> Mayo Clinic - Arizona Recruiting Phoenix, Arizona, United States, 85054 Contact: Clinical Trials Office 855-776-0015 Principal Investigator: Ruben Mesa, MD United States, Colorado University of Colorado Cancer Center Recruiting Aurora, Colorado, United States, 80045 Contact: Clinical Trials Office 720-848-0018 Principal Investigator: Daniel A Pollyea, MD, MS United States, Tennessee Vanderbilt-Ingram Cancer Center Recruiting Nashville, Tennessee, United States, 37232 Contact: Clinical Trials Office 800-811-8480 Principal Investigator: Michael R Savona, MD United States, Wisconsin Medical College of Wisconsin Recruiting Milwaukee, Wisconsin, United States, 53226 Contact: Clinical Trials Office 414-805-1118 Principal Investigator: Laura C. Michaelis, MD Collapse << |
| NCT03286530 | Acute Myeloid Leukemia ... more >> Acute Myeloid Leukemia in Remission Allogeneic Stem Cell Transplantation Collapse << | Phase 2 | Recruiting | September 1, 2024 | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Recruiting Boston, Massachusetts, United States, 02115 Contact: Myrna Nahas, MD 617-667-9920 Principal Investigator: Myrna Nahas, MD Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02115 Contact: Gabriella Hobbs, MD 617-726-8748 Principal Investigator: Gabriella Hobbs, MD United States, Missouri Washington University Recruiting Saint Louis, Missouri, United States, 63130 Contact: Mark Schroeder, MD markschroeder@wustl.edu Principal Investigator: Mark Schroeder, MD United States, Ohio The Ohio State University Recruiting Columbus, Ohio, United States, 43210 Contact: Sumithira Vasu, MBBS Sumithira.Vasu@osumc.edu Principal Investigator: Sumithira Vasu, MBBS United States, Tennessee Vanderbilt University Recruiting Nashville, Tennessee, United States, 37235 Contact: Michael Byrne, MD Michael.Byrne@vanderbilt.edu Principal Investigator: Michael Byrne, MD Collapse << |
| NCT03041636 | Malignant Neoplasms Stated as ... more >>Primary Lymphoid Haematopoietic Other Diseases of Blood and Blood-Forming Organs Chronic Lymphocytic Leukemia Small Lymphocytic Lymphoma Collapse << | Phase 2 | Recruiting | March 2021 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact zestrov@mdanderson.org Collapse << |
| NCT03427866 | Myelofibrosis | Phase 2 | Recruiting | September 30, 2021 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02214 Contact: Meridith E. Bailey, MD 617-643-4970 mebailey@partners.org Principal Investigator: Gabriela Hobbs, MD United States, Tennessee Vanderbilt University Recruiting Nashville, Tennessee, United States, 37235 Contact: Michael Byrne, DO Michael.Byrne@vanderbilt.edu Principal Investigator: Michael Byrne, DO Collapse << |
| NCT03722407 | Chronic Myelomonocytic Leukemi... more >>a Leukemia Collapse << | Phase 2 | Not yet recruiting | February 1, 2024 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Not yet recruiting Tampa, Florida, United States, 33612 Contact: Anthony McLaughlin 813-745-5941 Anthony.McLaughlin@moffitt.org Principal Investigator: Eric Padron, MD Sub-Investigator: Rami Komrokji, MD Sub-Investigator: Alan F List, MD Sub-Investigator: Jeffrey E Lancet, MD Sub-Investigator: Kendra Sweet, MD Sub-Investigator: Bijal Shah, MD Sub-Investigator: David Sallman, MD Sub-Investigator: Chetasi Talati, MD Sub-Investigator: Andrew Kuykendall, MD Sub-Investigator: Maria Balasis, BS Sub-Investigator: Brooke Fridley, PhD Collapse << |
| NCT02784496 | Myelofibrosis | Phase 2 | Active, not recruiting | September 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02928978 | Ductal Carcinoma In Situ ... more >> Atypical Lobular Hyperplasia Atypical Ductal Hyperplasia Lobular Carcinoma In Situ Collapse << | Phase 2 | Recruiting | January 2020 | United States, Texas ... more >> Baylor College of Medicine Recruiting Houston, Texas, United States, 77030 Contact: Kristen Otte 713-798-8874 clinical-research@breastcenter.tmc.edu Principal Investigator: Julie Nangia, M.D. Sub-Investigator: Mothaffar Rimawi, M.D. Sub-Investigator: Matthew Ellis, M.D. Sub-Investigator: Maryam Nemati Shaefee, M.D. Sub-Investigator: C. Kent Osborne, M.D. Sub-Investigator: Angela Coscio, M.D. Collapse << |
| NCT02912754 | Leukemia, Lymphocytic, Chronic... more >>, B-Cell Collapse << | Phase 1 Phase 2 | Not yet recruiting | August 2019 | Canada, Ontario ... more >> Sunnybrook Odette Cancer Center Not yet recruiting Toronto, Ontario, Canada, M4N 3M5 Contact: Geetha Geetha Yogendran, BSc 416-480-5000 ext 89565 geetha.yogendran@sunnybrook.ca Contact: Lindsay McCaw, MSc 416-480-6100 ext 4244 lindsay.mccaw@sunnybrook.ca Principal Investigator: david e spaner, MD Sunnybrook Odette Cancer Center Toronto, Ontario, Canada, M4N3M5 Collapse << |
| NCT02809976 | Vitiligo | Phase 2 | Completed | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT02041429 | Recurrent Breast Cancer ... more >> Metastatic Breast Cancer Collapse << | Phase 1 Phase 2 | Active, not recruiting | January 2021 | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| JAK1 | IC50:3.3nM | |
| JAK2 | IC50:2.8nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
